
09 Apr Private Label Cosmetics Manufacturing Process for Global Brands
Full Process of Private Label Cosmetics Manufacturing for Global Brands
For global beauty brands, retailers, and emerging entrepreneurs, private label cosmetics manufacturing is the fastest, lowest-risk way to build a unique product line, control profit margins, and enter the global beauty market. But many brands face costly launch delays, customs detention, product quality failures, and irreversible brand reputation damage, all from missing key details in the private label cosmetics manufacturing workflow. This guide breaks down the full end-to-end private label cosmetics manufacturing process into 8 actionable phases, with critical checkpoints, risk-avoidance tips, and compliance guidance to help you launch successfully. Read on to avoid common pitfalls and build a market-competitive beauty line.
📩Want to skip costly mistakes and launch your private label line on time? Use this complete private label cosmetics manufacturing workflow to lock in a trusted manufacturing partner and start your project today.
8 Core Phases of Private Label Cosmetics Manufacturing
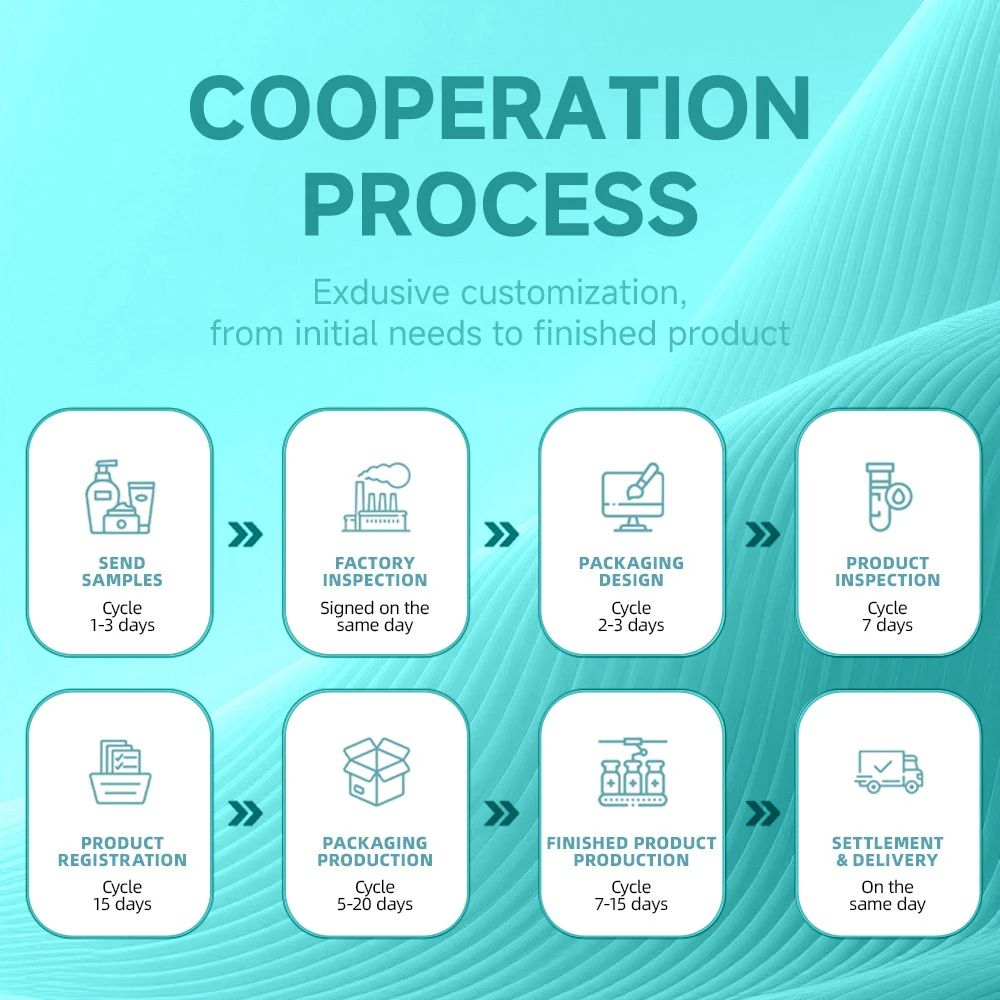
- Pre-Cooperation Planning for Your Private Label Cosmetics Manufacturing Project
This phase is the foundation of your entire private label cosmetics manufacturing project, directly determining if your final product aligns with your brand and market goals. Rushing this step is the #1 mistake new brands make.
Core tasks:
- Define your brand positioning, target market (EU/US/ASEAN/Middle East), core consumer groups, product category, efficacy claims, budget, MOQ, and launch timeline
- Confirm target market cosmetic regulations (ingredient restrictions, label rules, efficacy claim limits) with a professional private label cosmetics manufacturingpartner
- Screen qualified manufacturers with GMPC/ISO 22716 certifications and relevant export experience, and confirm the basic cooperation framework
- Sign a comprehensive Non-Disclosure Agreement (NDA) before sharing any brand confidential information
- Formula R&D for Your Private Label Cosmetics Manufacturing Line
Formula is the core competitiveness of your products, and this phase defines the efficacy, user experience, and safety of your final private label cosmetics manufacturing line.
Core workflow:
- Customize exclusive formulas based on your market needs (e.g. clean beauty/vegan formulas for EU/US, oil-control formulas for Southeast Asia), or select and adjust mature market-verified formulas
- Produce prototype samples for your full testing (texture, skin feel, fragrance, absorption, efficacy experience)
- Complete multi-round revisions until 100% satisfaction, then seal duplicate samples as the only quality standard for mass production
- Conduct pre-production formula safety and stability testing (high/low temperature cycle, microbial challenge, skin irritation test)
- Packaging & Compliance for Private Label Cosmetics Manufacturing
Over 60% of cosmetic export customs detentions stem from non-compliant packaging labels, making this a critical risk-mitigation phase for your private label cosmetics manufacturing project.
Core tasks:
- Confirm packaging type, material, structure, and craftsmanship, with professional guidance from your manufacturer to ensure compatibility with mass production equipment
- Complete packaging graphic design following the manufacturer’s standard specs, including brand visuals and mandatory information placement
- Full regulatory compliance review of all label content (ingredient list order, efficacy claims, net content, manufacturer info, warning language) by the manufacturer’s regulatory team
- Produce and confirm packaging samples, then seal them as the mass production standard
- Pre-Production Preparation for Private Label Cosmetics Manufacturing
This phase links R&D to mass production, ensuring smooth execution of your private label cosmetics manufacturing project and avoiding delays from supply chain issues.
Core workflow:
- Sign a formal cooperation contract with clear product specs, order quantity, pricing, production cycle, quality standards, payment terms, and liability clauses
- Pay the deposit to initiate raw material and packaging procurement from long-term, high-quality suppliers
- Complete strict Incoming Quality Control (IQC) for all materials, only qualified materials can enter production
- Conduct a small-batch pre-production trial run to verify process stability and packaging compatibility, resolve issues before formal mass production
- Mass Production & QC for Private Label Cosmetics Manufacturing
This phase turns your product vision into tangible finished goods, with strict GMP-standard controls to ensure consistent quality across every batch of your private label cosmetics manufacturing line.
Core control points:
- All production completed in a 100,000-level dust-free GMP clean workshop, compliant with GMPC/ISO 22716 international standards (learn more about ISO 22716 requirements from the official ISO website)
- Strict production following sealed formulas and Standard Operating Procedures (SOP) for emulsification, filling, capping, coding, and packaging
- Real-time In-Process Quality Control (IPQC) monitoring and recording for every production link
- Full batch traceability system with complete records of raw material batches, production parameters, operators, and quality inspection logs
- Finished Product Inspection for Private Label Cosmetics Manufacturing
This is the final quality and compliance checkpoint before products leave the factory for your private label cosmetics manufacturing project.
Core tasks:
- Comprehensive Finished Quality Control (FQC) for every batch, including microbial testing, heavy metal screening, ingredient content verification, stability testing, and packaging/label inspection
- Optional third-party testing reports from authoritative institutions (SGS, Intertek) per your market requirements
- Preparation of a full set of compliance and export documents: Certificate of Analysis (COA), Safety Data Sheet (SDS), Product Information File (PIF), Certificate of Origin, and more
- Only fully qualified, document-complete batches can be officially released for delivery
- Logistics & Customs Support for Private Label Cosmetics Manufacturing
A professional private label cosmetics manufacturing partner provides end-to-end support, not just factory-gate delivery, to ensure your products reach your target market smoothly.
Core services:
- Customized logistics solutions (sea freight, air freight, international express) balancing cost and timeliness
- One-stop export customs clearance service with professional cosmetic export agencies
- Full cargo tracking and real-time logistics status updates
- Safe on-time delivery to your designated warehouse/overseas warehouse, with full support for the acceptance process
- After-Sales Support for Ongoing Private Label Cosmetics Manufacturing
A trusted private label cosmetics manufacturing partner is a long-term strategic asset that grows with your brand, not just a one-time supplier.
Core long-term support:
- 24-hour rapid after-sales response with clear solutions for any product feedback
- Priority production scheduling for replenishment orders to avoid stockouts, especially during peak sales seasons
- Ongoing formula upgrade and new product development support aligned with global beauty market trends
- Real-time regulatory update alerts and adjustment guidance for your target markets (learn more about US cosmetic compliance rules from the U.S. FDA official website)
Quick Comparison: Standard vs. Professional Private Label Cosmetics Manufacturing Partner
| Evaluation Item | Standard Manufacturer | Professional Private Label Cosmetics Manufacturing Partner |
|---|---|---|
| Core Certifications | Basic local certificates only | GMPC, ISO 22716, FDA, EU CPNP compliant |
| R&D Capability | Basic fixed formulas only | Custom formula development, sample sealing, full safety testing |
| Quality Control | Simple final inspection only | Full IQC-IPQC-FQC system with end-to-end batch traceability |
| Compliance Support | Limited export documents only | Full regulatory review, export docs, and global market registration support |
| MOQ Policy | High fixed MOQ only | Flexible low MOQ (1000-3000 units/SKU) for startup brands |
| After-Sales Support | Slow, limited response | 24h response, replenishment support, and long-term product iteration |
Key Pitfall Avoidance Tips for Private Label Cosmetics Manufacturing
- Prioritize private label cosmetics manufacturingpartner qualifications and export experience over ultra-low prices, which often mean inferior materials and non-compliant production
- Confirm target market regulatory requirements at the very start of your private label cosmetics manufacturingproject to avoid costly rework
- Strictly implement sample sealing for all formulas and packaging, as the only standard for mass production and acceptance
- Clearly define intellectual property ownership in the formal contract, and sign an NDA before any confidential information sharing
FAQ: Private Label Cosmetics Manufacturing
Q1: How long does the full private label cosmetics manufacturing process take?
A1: The standard full cycle is 4-8 weeks. Mature formulas with conventional packaging can be completed in 4-6 weeks, while fully customized formulas and custom mold packaging take 6-8 weeks. Professional private label cosmetics manufacturing partners provide a clear timeline and deliver strictly on schedule.
Q2: What is the typical MOQ for private label cosmetics manufacturing?
A2: For conventional skincare products (cleanser, serum, moisturizer), professional private label cosmetics manufacturing partners offer a startup-friendly MOQ of 1000-3000 units per SKU, with flexible higher volumes for established brands. MOQ for custom packaging will be confirmed upfront based on supplier requirements.
Q3: Who owns the intellectual property rights of the private label formula and packaging design?
A3: Brand-created packaging designs and proprietary formulas fully belong to your brand. For manufacturer-provided mature formulas, your brand gets exclusive use rights. For fully customized exclusive formulas for your private label cosmetics manufacturing project, full IP ownership can be defined in the contract.
Q4: Can a private label cosmetics factory help with product registration in our target market?
A4: Yes. Professional private label cosmetics manufacturing partners with global export experience provide full registration support, including all required compliance documents, and assist with EU CPNP notification, US FDA VCRP filing, and other market access procedures.






No Comments